Ozempic, Wegovy, Mounjaro, and Zepbound:
Understanding the Differences and Finding the Right Path Forward
Few topics in modern medicine have generated as much public interest and confusion as the new generation of injectable weight-loss medications.
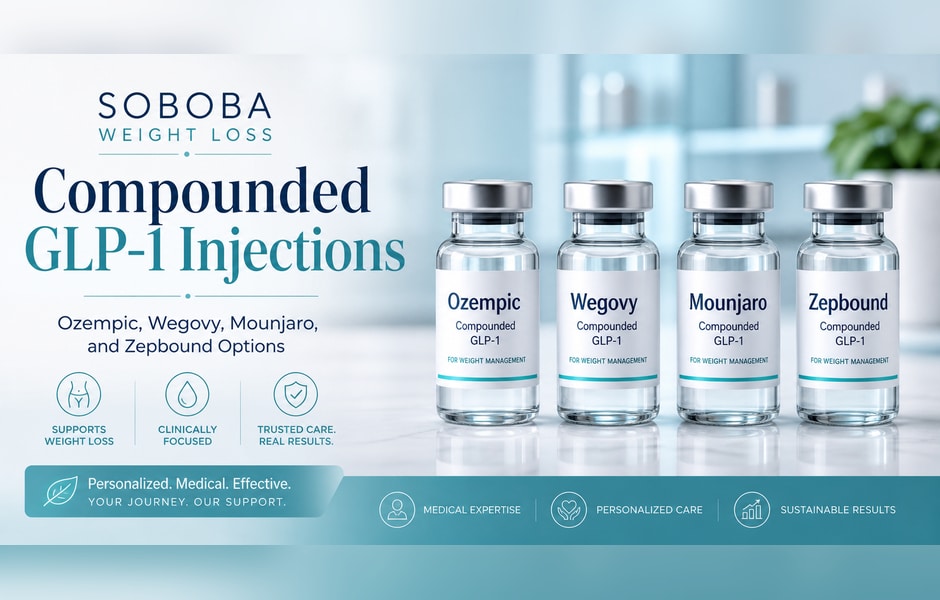
Names like Ozempic, Wegovy, Mounjaro, and Zepbound now appear constantly in news headlines, social media feeds, and dinner-table conversations. Celebrities have endorsed them. Shortages have made pharmacies scramble. And millions of Americans are asking their doctors whether one of these drugs might be right for them. But despite the buzz, many people remain unclear about what these medications actually are, how they differ from one another, and what separates a safe, supervised treatment plan from a dangerous shortcut. This essay explains the science behind each drug, the key distinctions that matter most, and why working with a qualified medical weight-loss clinic such as Soboba Weight Loss Clinics in Orange County, California, is the safest and most effective path to lasting results.
The GLP-1 Revolution: A New Class of Medications
All four medications, Ozempic, Wegovy, Mounjaro, and Zepbound, belong to a class of drugs that mimic naturally occurring hormones in the body that regulate blood sugar and appetite. The most important of these hormones is called glucagon-like peptide-1, or GLP-1, which is released by the gut after eating. GLP-1 tells the pancreas to release insulin, slows the rate at which the stomach empties food into the intestine, and signals the brain to produce feelings of fullness and satiety. For people who struggle with obesity, this appetite-regulation system is often impaired. GLP-1 receptor agonist drugs that activate GLP-1 receptors in the body have proven to be a breakthrough in restoring that regulation.
Two of these medications, semaglutide and tirzepatide, are the active ingredients behind all four brand names currently dominating the conversation. Understanding the difference between these two molecules is the first key to understanding the drugs themselves.
Ozempic and Wegovy: Two Brands, One Molecule
Ozempic and Wegovy are both brand names for the same drug: semaglutide, manufactured by the Danish pharmaceutical company Novo Nordisk. Semaglutide is a GLP-1 receptor agonist. It mimics the GLP-1 hormone to regulate blood sugar, slow gastric emptying, and reduce appetite. Yet the two brand names exist for a very specific reason: they are approved by the U.S. Food and Drug Administration (FDA) for different conditions and come in different dosage strengths.
Ozempic received FDA approval in 2017 specifically for the management of type 2 diabetes in adults. It helps lower blood sugar levels and has the added benefit of reducing the risk of major cardiovascular events such as heart attacks and strokes in people with diabetes and heart disease. While many patients on Ozempic do lose significant weight as a side effect, the drug is not officially approved for weight loss. The maximum approved dose of Ozempic is 2 mg weekly.

Wegovy, on the other hand, received FDA approval in 2021 specifically for chronic weight management in adults with obesity (a BMI of 30 or higher) or in adults who are overweight (a BMI of 27 or higher) with at least one weight-related health condition such as high blood pressure, type 2 diabetes, or high cholesterol. Wegovy is dosed at a higher dose than Ozempic, up to 2.4 mg weekly( That is correct for the standard injectable Wegovy maintenance dose, as of March 2026, the current the FDA approved a newer Wegovy HD 7.2 mg), and clinical trials showed that participants lost an average of about 15% of their body weight over 68 weeks, a result unprecedented in the history of pharmaceutical weight management. On March 8, 2024, the FDA also approved Wegovy to reduce the risk of serious cardiovascular events in adults with established heart disease and obesity or overweight.
The key distinction between Ozempic and Wegovy, then, lies not in the molecule but in their intended use and dose. Prescribing Ozempic for weight loss when a patient does not have type 2 diabetes is considered an “off-label” use, which is legal but not FDA-indicated. Wegovy is the appropriate choice when the primary goal is weight management.
Mounjaro and Zepbound: The Dual-Action Breakthrough
Mounjaro and Zepbound are both brand names for tirzepatide, a medication manufactured by Eli Lilly. Tirzepatide represents a meaningful advance beyond semaglutide because it targets not one but two hormonal pathways simultaneously. In addition to activating GLP-1 receptors, tirzepatide also activates receptors for glucose-dependent insulinotropic polypeptide, or GIP, another gut hormone involved in blood sugar regulation and fat metabolism. This dual mechanism of action is why tirzepatide is classified as a “GIP/GLP-1 receptor agonist” rather than simply a GLP-1 receptor agonist.
Mounjaro received FDA approval in 2022 for the treatment of type 2 diabetes in adults. Like Ozempic, it was approved for its glucose-lowering effects, and weight loss emerged as a notable secondary benefit in clinical trials. Patients using Mounjaro for diabetes management often experienced substantial weight loss, setting the stage for the drug’s next chapter.

Zepbound received FDA approval in November 2023 specifically for chronic weight management, mirroring the relationship between Ozempic and Wegovy. Clinical trials for Zepbound were striking: participants lost an average of approximately 20% of their body weight over 72 weeks at the highest dose, outcomes that exceeded those seen with semaglutide alone. At lower doses, the average weight loss was still in the 15% range, which remains a remarkable figure by historical standards. Zepbound has since also received approval for obstructive sleep apnea in adults with obesity, expanding its clinical applications.
Just as with Ozempic and Wegovy, the distinction between Mounjaro and Zepbound is primarily one of indication and approved use. Mounjaro is prescribed for type 2 diabetes; Zepbound is the weight-management version of the same molecule. The dual-action mechanism of tirzepatide — engaging both GIP and GLP-1 receptors — appears to deliver stronger results than GLP-1 activation alone, which is why many clinicians and researchers view tirzepatide as the current leading option for patients who are candidates for either drug.
Side Effects, Considerations, and Who Qualifies
All four medications share a common side-effect profile rooted in their mechanism of slowing gastric emptying. Nausea, vomiting, diarrhea, constipation, and abdominal discomfort are the most commonly reported issues, particularly in the early weeks of treatment as the dose is gradually increased. These side effects are usually temporary and tend to diminish as the body adjusts. More serious but rare risks include pancreatitis, gallbladder disease, and, in animal studies only, thyroid tumors. All drugs in this class carry a boxed warning against use in patients with a personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2.

These medications are not intended for cosmetic weight loss in people who are simply looking to shed a few pounds. FDA approval criteria require a BMI of 30 or above, or 27 or above with a qualifying comorbidity. Nor are they magic solutions; the research is detailed: weight tends to return if the medications are stopped without accompanying lifestyle changes. They are tools, not cures, and they work best as part of a comprehensive program that includes medical supervision, nutritional guidance, behavioral support, and physical activity.
Why Medical Supervision Matters: Soboba Weight Loss Clinics in Orange County
The proliferation of online pharmacies and telehealth platforms offering GLP-1 medications with minimal evaluation has raised real concerns in the medical community. While convenient access to medication sounds appealing, weight loss is a complex medical endeavor. Prescribing semaglutide or tirzepatide without a thorough evaluation of a patient’s health history, current medications, metabolic profile, and personal goals is not just substandard care — it can be genuinely dangerous. Drug interactions, contraindications, and the need to manage side effects all require ongoing, personalized medical oversight.
This is precisely where Soboba Weight Loss Clinics distinguishes itself for residents of Orange County and the broader Southern California region. Soboba operates as a medically supervised weight loss program with clinical staff who understand that sustainable weight management requires far more than a prescription. Patients at Soboba receive comprehensive initial evaluations that assess their medical history, laboratory values, body composition, and weight-loss goals. From that foundation, a personalized treatment plan is developed — one that may include GLP-1 medications where appropriate, but always integrated into a broader strategy.
What sets medically supervised programs apart is the continuity of care. When side effects arise, a qualified medical provider is available to adjust dosing, recommend strategies for managing nausea or GI discomfort, and determine when it is appropriate to slow or accelerate a patient’s titration schedule. When a patient has other health conditions, such as high blood pressure, pre-diabetes, hypothyroidism, or a history of eating disorders, those factors must inform the treatment plan. Soboba’s clinical approach ensures that these nuances are accounted for at every step.
Beyond medication management, Soboba integrates nutritional counseling, behavioral coaching, and accountability check-ins, all of which are essential for long-term success. Research on GLP-1 medications consistently shows that patients who combine pharmacotherapy with lifestyle interventions achieve better outcomes and are better positioned to maintain those results over time. A clinic like Soboba creates the structure for that integration, turning a medication into the foundation of a genuine lifestyle transformation rather than a temporary fix.

For Orange County residents who have struggled with weight for years, who have tried diets, exercise programs, and other interventions without lasting success, the combination of today’s remarkable medications and Soboba’s expert medical guidance represents a genuinely new possibility. Whether the right medication is Wegovy or Zepbound, or whether a patient’s situation calls for a different approach entirely, those decisions deserve the attention of trained professionals who see the full picture of a patient’s health.
Conclusion: The Right Drug, the Right Guidance
Ozempic, Wegovy, Mounjaro, and Zepbound are not interchangeable names for the same thing. They represent two distinct molecules, semaglutide and tirzepatide, each available in two brand versions targeting different FDA-approved indications. Ozempic and Mounjaro are approved for type 2 diabetes management; Wegovy and Zepbound are approved for chronic weight management. Tirzepatide’s dual GIP/GLP-1 mechanism appears to produce greater average weight loss than semaglutide alone, though both are transformative by historical standards. All four share similar side effects, similar contraindications, and a shared requirement: they work best when guided by medical expertise.
For anyone in Orange County, California,s considering whether one of these medications might be appropriate for them, the most important first step is not to search for the cheapest online pharmacy or the fastest shipping option. The most important step is to consult with a qualified medical team. Soboba Weight Loss Clinics offer that expertise, that continuity of care, and that commitment to treating the whole person, not just the number on a scale. In a landscape crowded with marketing hype and shortcut solutions, that kind of rigorous, compassionate medical oversight remains the difference between short-term results and long-term health.

For patients comparing today’s leading medical weight-loss options, Soboba Weight Loss offers helpful information about Ozempic Injections in Orange County, Mounjaro Injections in Orange County, and the Wegovy Pill. These treatments may support appetite control, blood sugar regulation, and long-term weight management when used under proper medical supervision. Patients can also review current GLP-1 Specials to learn more about available offers and treatment options.
Soboba Weight Loss Clinics in Orange County provides medically supervised weight loss care for patients throughout the region. Whether you are looking for a Laguna Hills medical weight loss clinic, a Newport Beach medical weight loss clinic, or a Rancho Santa Margarita medical weight loss clinic, Soboba Weight Loss can help you choose a safe and personalized path toward your weight loss goals.